Phenol vs Phenyl: Understanding the Essential Differences
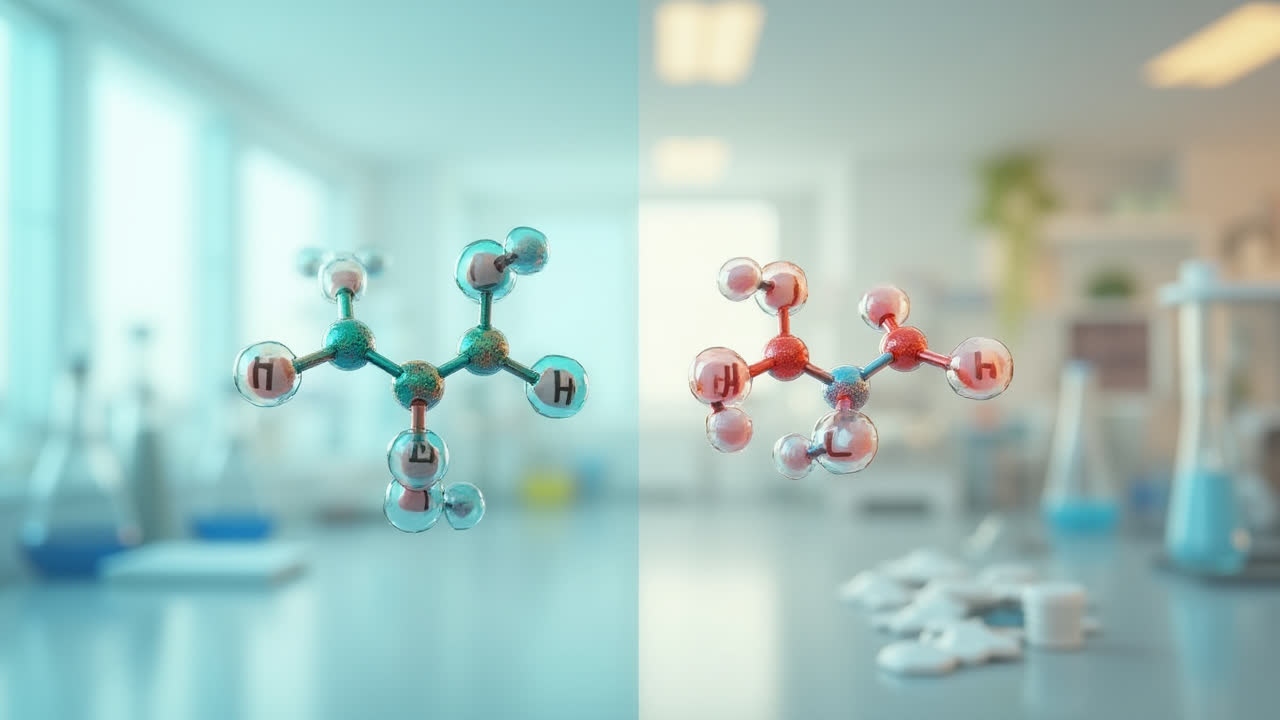
Chemistry students and professionals alike often confuse phenol and phenyl due to their similar names and related chemical structures. I remember my own struggles during organic chemistry labs, mixing up these terms until a patient professor set me straight. These two chemical entities may sound alike, but they serve entirely different purposes in the world of chemistry. Let's dive into what makes each unique and why knowing the difference matters.
In the simplest terms, phenol is a complete, stable compound with a hydroxyl group attached to a benzene ring, while phenyl is an unstable functional group derived from benzene by removing one hydrogen atom. This distinction might seem minor at first glance, but it leads to vastly different chemical behaviors and applications that impact everything from pharmaceutical development to materials science.
What is Phenol? Structure and Properties
Phenol is an aromatic organic compound with the molecular formula C6H5OH. It consists of a benzene ring with a hydroxyl group (-OH) directly attached to it. This structural arrangement makes phenol the simplest member of the class of compounds known as aromatic alcohols. When I first encountered phenol in the lab, I was struck by its distinctive sweet yet medicinal odor—something you definitely don't forget!
At room temperature, phenol appears as a crystalline solid with a transparent appearance. It has a melting point of approximately 40.5°C and a boiling point around 181.7°C. One interesting property of phenol is its ability to mix with water due to hydrogen bonding between the hydroxyl group and water molecules. However, don't let this water-friendly characteristic fool you—phenol is actually quite acidic compared to other alcohols. Why? Because the negative charge that forms when phenol loses a proton gets stabilized through resonance across the benzene ring.
The acidity of phenol explains why it partially dissociates in aqueous solutions to form phenolate ions and hydronium cations. This property makes phenol a weak acid with a pKa value of about 10. In practical terms, this means phenol is about a million times more acidic than typical alcohols like ethanol. Isn't that remarkable? A single structural difference creating such a dramatic change in chemical behavior!
Phenol's stability comes from its complete electron configuration and resonance stabilization. Unlike its cousin phenyl, phenol doesn't actively seek to react with other compounds (though it certainly can under the right conditions). This stability, combined with its unique properties, makes phenol incredibly valuable for numerous applications across different industries.
Common Applications of Phenol
You might be surprised by how often you encounter phenol in your daily life. Despite its toxicity—and yes, phenol is definitely toxic and corrosive to eyes and skin—it serves as a crucial building block in modern industry. Back in the 19th century, Joseph Lister revolutionized surgical practices by using phenol as an antiseptic. Though we've developed safer alternatives since then, this historic application highlights phenol's powerful antimicrobial properties.
Today, phenol serves as a precursor for numerous products we use regularly. The majority of phenol production goes toward making plastics, particularly phenolic resins used in plywood adhesives and the manufacturing of circuit boards. Have you ever wondered what gives some mouthwashes their distinctive taste? Often, it's dilute phenol providing that medicinal kick while fighting oral bacteria.
Phenol derivatives also play crucial roles in pharmaceutical development. Acetylsalicylic acid—better known as aspirin—is perhaps the most famous example of a medication derived from phenolic compounds. Other examples include tyrosol, found in olive oil, which has antioxidant properties, and capsaicin, the compound responsible for the heat in chili peppers that's now used in pain-relieving creams.
What is Phenyl? Structure and Reactivity
Unlike phenol, phenyl is not a complete compound but rather a functional group with the formula C6H5-. Think of it as a benzene ring that's missing one hydrogen atom, creating a reactive site where the ring can attach to other chemical structures. This seemingly small difference—just one hydrogen atom—completely changes how it behaves chemically.
The phenyl group features a planar, cyclic structure where five carbon atoms bond to five hydrogen atoms individually, while the sixth carbon atom has an available bonding site. This vacant spot makes the phenyl group highly reactive—it's essentially looking for an electron to complete its configuration. I like to think of the phenyl group as a chemical "hook" that can grab onto other molecules and incorporate them into larger structures.
One fascinating aspect of the phenyl group is its enhanced stability compared to aliphatic alkyl groups of similar size. This stability comes from the delocalization of pi electrons throughout the aromatic ring system. However, don't confuse this relative stability with being stable on its own—a lone phenyl group is actually quite reactive and eager to form bonds.
In organic synthesis, phenyl groups often serve as important structural components that can influence a molecule's physical properties, reactivity, and biological activity. Chemists frequently incorporate phenyl groups into drug molecules to improve their binding to biological targets or to modify their solubility. When I was working on a research project, we added phenyl groups to several candidate molecules and found dramatic improvements in their ability to cross cell membranes—a perfect example of how this simple structure can have profound effects.
Phenyl vs Phenol: Comparison Table
| Feature | Phenol | Phenyl |
|---|---|---|
| Chemical Type | Complete compound | Functional group |
| Molecular Formula | C6H5OH | C6H5- |
| Structure | Benzene ring with hydroxyl group | Benzene ring missing one hydrogen |
| Stability | Stable compound | Unstable alone, reactive |
| Oxygen Content | Contains oxygen atom | No oxygen atoms |
| Physical State | Crystalline solid at room temperature | Not found alone in nature |
| Reactivity | Less reactive, stable structure | Highly reactive, seeks electron-sharing |
| Primary Use | Precursor for plastics, disinfectant | Building block in organic synthesis |
Key Molecular Differences
The fundamental difference between phenol and phenyl lies in their molecular composition and completeness. Phenol contains an oxygen atom as part of its hydroxyl group, while phenyl lacks any oxygen atoms entirely. This distinction might seem straightforward, but it underlies virtually all the other differences between these two chemical entities.
Looking at electronic structure, phenol has a complete electron configuration, with all atoms having their valence shells filled through bonding. The phenyl group, by contrast, has one carbon atom with an incomplete valence shell—it's essentially looking for something to bond with to achieve stability. This electronic incompleteness is why you'll never find phenyl groups floating around independently in nature.
Another interesting distinction relates to acidity. The presence of the hydroxyl group makes phenol weakly acidic, allowing it to donate a proton in certain reactions. The phenyl group, lacking this hydroxyl feature, doesn't display acidic properties. Instead, its primary chemical characteristic is its ability to attach to other molecular structures through the formation of covalent bonds.
From a synthetic chemistry perspective, these differences mean that phenol and phenyl serve completely different roles. Phenol functions as a starting material or reagent in reactions, while phenyl acts as a structural component that gets incorporated into larger molecules. I've worked with both in different contexts—using phenol as a reactant to produce more complex compounds, and attaching phenyl groups to enhance the properties of pharmaceutical candidates.
Common Confusion Points
Why do so many chemistry students (and sometimes even professionals) confuse these two terms? Beyond their similar spellings, there's actually a logical connection between them: phenol contains a phenyl group linked to a hydroxyl group. You could say that phenol is a hydroxylated phenyl derivative. This relationship creates a conceptual overlap that can lead to confusion.
Another source of mix-ups comes from naming conventions. Many phenol derivatives are named using "phenyl" in their nomenclature. For example, 2-phenylethanol contains a phenyl group but is not the same as phenol. These nomenclature subtleties require close attention, especially when first learning organic chemistry.
I remember a lab partner who consistently wrote "phenyl alcohol" when they meant phenol. While not entirely incorrect (phenol could be described as a phenyl alcohol), this imprecise terminology created confusion in our reports. Chemistry demands precision in language because small differences in terms often reflect critical differences in molecular structure and behavior.
FAQ: Phenol vs Phenyl
No, phenyl cannot exist independently in nature. As a functional group with an incomplete electron configuration, it is inherently unstable on its own. The phenyl group always needs to be attached to other atoms or groups to achieve stability. This is why you'll only ever encounter phenyl as part of larger molecular structures like alkylbenzenes, phenyl ethers, or phenol itself (which is a phenyl group attached to a hydroxyl group).
Yes, phenol requires careful handling in laboratory settings. It's corrosive to eyes and skin, causing severe burns upon contact, and its vapors can irritate the respiratory system. Phenol can also be absorbed through the skin, potentially causing systemic toxicity. When working with phenol, always use appropriate personal protective equipment including gloves (nitrile or butyl rubber), eye protection, and a lab coat. Work should be conducted in a well-ventilated area or fume hood. Despite these hazards, phenol remains widely used in controlled laboratory environments due to its valuable properties and applications.
You can identify compounds containing phenyl groups through several methods. In chemical names, look for the term "phenyl" or "Ph" abbreviation (like in Ph-CH3 for toluene). In structural formulas, look for a hexagonal ring with alternating double bonds (or the simplified notation of a hexagon with a circle inside) that's attached to another structure. Spectroscopically, compounds with phenyl groups show characteristic patterns in NMR spectra (typically signals between 7-8 ppm for aromatic protons) and distinctive absorption bands in IR spectroscopy around 1600 and 1450 cm-1. These aromatic compounds also typically have higher boiling points than their non-aromatic counterparts.
Conclusion
Understanding the distinction between phenol and phenyl is fundamental to grasping organic chemistry concepts and nomenclature. While they share a common structural feature—the benzene ring—their differences in completeness, stability, and reactivity make them distinct chemical entities with unique applications and behaviors.
Phenol, with its hydroxyl group, functions as a complete, stable compound that serves as an important industrial chemical and precursor to numerous useful products. The phenyl group, by contrast, acts as a molecular building block that gets incorporated into more complex structures to modify their properties.
Next time you encounter these terms in your chemistry studies or work, you'll be able to distinguish between them confidently. And if you ever find yourself confusing them again (as even experienced chemists sometimes do), just remember: phenol is a complete compound with an oxygen-containing hydroxyl group, while phenyl is an incomplete fragment eager to form new bonds. This simple distinction will serve you well in your chemical journey.