Understanding Pyknosis, Karyolysis, and Karyorrhexis: Key Differences
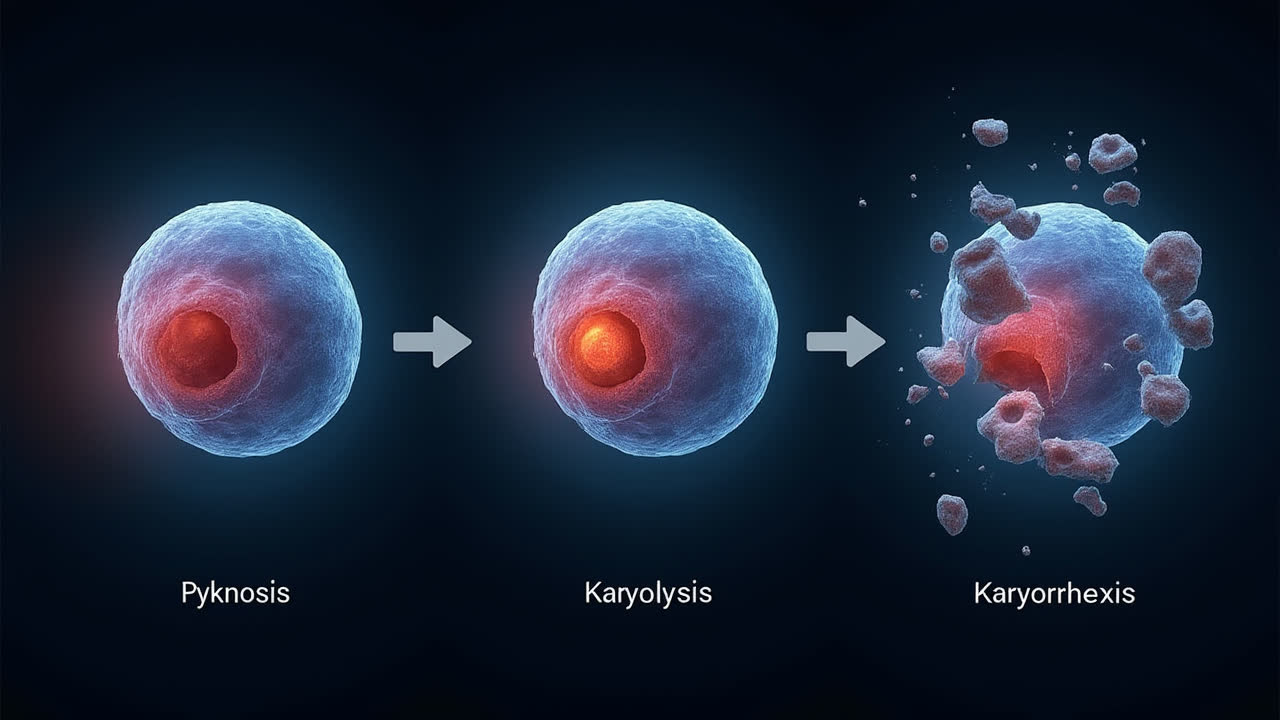
Understanding Pyknosis, Karyolysis, and Karyorrhexis: Key Differences
When cells begin to die, whether through programmed cell death (apoptosis) or accidental cell death (necrosis), they undergo specific morphological changes in their nuclei. Pyknosis, karyolysis, and karyorrhexis represent three distinct nuclear degradation processes that occur during cell death. While these terms might sound intimidatingly technical, understanding their differences provides fascinating insights into how our cells break down when they're damaged beyond repair or when they've reached the end of their natural lifecycle.
Have you ever wondered what actually happens inside a cell when it dies? I certainly have. The nucleus – that command center housing our precious DNA – doesn't just disappear. Instead, it undergoes a series of remarkable transformations. Sometimes I think of it like watching a building being demolished in slow motion – there's a method to the breakdown, with distinct stages that cellular pathologists can identify under the microscope.
What is Pyknosis?
Pyknosis represents the first major morphological change observed in a dying cell's nucleus. It involves the irreversible condensation of chromatin, causing the nucleus to shrink and become more densely stained when viewed under a microscope. During this process, the genetic material (chromatin) becomes increasingly compact and deeply colored when stained with histological dyes.
Interestingly, pyknosis manifests differently depending on whether the cell is dying through apoptosis or necrosis. In apoptotic cells, the chromatin condenses into large, well-defined clumps that eventually get packaged into apoptotic bodies. Necrotic cells, on the other hand, show irregular, dispersed chromatin clumps that eventually dissolve. This distinctive appearance makes pyknosis a valuable marker for identifying dying cells in both laboratory cultures and tissue samples.
Beyond cell death scenarios, pyknosis also plays a role in normal cellular development. For example, it occurs during the maturation of red blood cells as they prepare to expel their nuclei completely. Similarly, neutrophils (a type of white blood cell) undergo a form of pyknosis where the nucleus condenses into connected lobes – a hallmark feature that helps pathologists identify these important immune cells.
What is Karyolysis?
Karyolysis represents a more advanced stage of nuclear degradation characterized by the complete dissolution of the nucleus. During this process, nuclear components gradually fade away due to enzymatic degradation by specialized proteins called endonucleases. DNase γ, a secreting protein of the DNase I family, plays a particularly important role in this process by facilitating nuclear fragmentation.
When a cell undergoes karyolysis, its entire nuclear structure eventually disappears, leaving the cytoplasm to stain uniformly with eosin (a common histological dye). This uniform staining pattern provides another visual clue for pathologists examining tissue samples. Karyolysis typically follows karyorrhexis in the sequence of cellular degradation events.
In certain diagnostic contexts, identifying karyolysis can provide valuable clinical information. For instance, malignant cells found in urine samples often display pyknosis and karyolysis of their nuclei. These nuclear changes, when properly identified, help pathologists distinguish between normal cellular turnover and potentially concerning abnormalities that might indicate disease.
What is Karyorrhexis?
Karyorrhexis describes the destructive fragmentation of a dying cell's nucleus. During this process, the nuclear membrane breaks down, and chromatin becomes distributed throughout the cytoplasm in an irregular pattern. This scattered appearance of nuclear material creates a distinctive visual signature that pathologists use to identify cells in this particular stage of degradation.
Occurring after pyknosis but before karyolysis, karyorrhexis represents an intermediate stage in nuclear breakdown. It can result from several cellular fates including programmed cell death, necrosis, or cellular senescence (the process of cells aging and losing their ability to divide). The irregular distribution of chromatin throughout the cytoplasm creates a distinctive appearance that helps distinguish karyorrhexis from other nuclear changes.
Karyorrhexis plays an especially important role in apoptosis, the programmed cell death mechanism that helps maintain tissue homeostasis throughout the body. This controlled demolition process is crucial for normal development, as it helps shape organs during embryogenesis and maintains proper cellular turnover in mature tissues. When karyorrhexis occurs as part of apoptosis, it represents a tightly regulated step in a healthy physiological process.
Pyknosis, Karyolysis, and Karyorrhexis: A Detailed Comparison
| Feature | Pyknosis | Karyolysis | Karyorrhexis |
|---|---|---|---|
| Definition | Irreversible condensation of chromatin with nuclear shrinkage | Complete dissolution of nuclear components | Destructive fragmentation of the nucleus |
| Primary Effect | Chromatin condensation and hyperchromatism | Nuclear fading and eventual disappearance | Nuclear fragmentation with irregular chromatin distribution |
| Sequence in Cell Death | Typically occurs first | Typically occurs last | Occurs after pyknosis but before karyolysis |
| Microscopic Appearance | Shrunken, deeply stained nucleus | Fading nucleus with uniform cytoplasmic staining | Fragmented nuclear material scattered in cytoplasm |
| Biological Mechanism | Chromatin compaction and marginalization | Enzymatic degradation by endonucleases (e.g., DNase γ) | Nuclear membrane breakdown |
| Cell Death Association | Occurs in both apoptosis and necrosis with different patterns | More commonly associated with necrosis | Can occur in apoptosis, necrosis, or cellular senescence |
| Non-Death Roles | Red blood cell and neutrophil maturation | Rarely occurs outside of cell death | Part of normal tissue remodeling during development |
| Diagnostic Value | Cell death marker in vivo and in vitro | Indicator of complete nuclear destruction | Marker for intermediate stage of cell death |
Biological Significance and Clinical Importance
Understanding the differences between pyknosis, karyolysis, and karyorrhexis extends beyond mere academic interest – these processes have significant implications for both normal physiology and pathological conditions. In normal development, programmed cell death helps shape tissues and organs, remove unnecessary structures, and maintain cellular homeostasis. The ability to identify and distinguish between different stages of nuclear degradation allows researchers and clinicians to assess cellular health with greater precision.
In clinical pathology, recognizing these nuclear changes provides valuable diagnostic information. For example, increased rates of cell death with characteristic nuclear changes might indicate tissue damage, inflammation, or malignant transformation. Pathologists routinely examine tissue samples for these nuclear alterations when assessing various disease states, from inflammatory conditions to cancer.
The study of these processes also informs therapeutic approaches. Many cancer treatments, for instance, work by inducing apoptosis in malignant cells. Understanding the mechanisms and markers of different cell death pathways helps researchers develop and assess the effectiveness of such treatments. Similarly, in degenerative diseases characterized by excessive cell death, identifying the specific pathways involved might reveal potential targets for intervention.
Frequently Asked Questions
The typical sequence of nuclear changes during cell death begins with pyknosis (irreversible chromatin condensation), followed by karyorrhexis (nuclear fragmentation), and finally karyolysis (complete dissolution of the nucleus). This sequence can vary somewhat depending on the specific type of cell death (apoptosis vs. necrosis) and the cell type involved, but this represents the general progression observed in most cases.
No, pyknosis, karyorrhexis, and karyolysis represent irreversible changes in nuclear morphology. Once a cell reaches these stages of nuclear degradation, it is committed to death. These processes involve fundamental breakdown of nuclear structures and DNA that cannot be repaired or reversed. This irreversibility makes these changes reliable markers for identifying cells that have passed the "point of no return" in the cell death process.
Pathologists distinguish between pyknosis, karyorrhexis, and karyolysis primarily through histological staining and microscopic examination. Pyknotic nuclei appear shrunken and deeply stained (hyperchromatic). Karyorrhectic nuclei show fragmentation with chromatin scattered throughout the cytoplasm. Karyolytic nuclei progressively fade and eventually disappear, leaving the cytoplasm to stain uniformly. Special stains that highlight DNA, such as hematoxylin, are particularly useful for identifying these different stages of nuclear degradation in tissue samples.
Conclusion
Pyknosis, karyolysis, and karyorrhexis represent three distinct processes in the nuclear degradation sequence during cell death. While they share a common association with cellular demise, each process creates distinct morphological changes that serve as important markers in both research and clinical contexts. Pyknosis involves the irreversible condensation of chromatin, karyorrhexis entails nuclear fragmentation, and karyolysis results in the complete dissolution of nuclear components.
These processes occur in a typical sequence during cell death, with pyknosis generally happening first, followed by karyorrhexis, and concluding with karyolysis. However, variations in this pattern can occur depending on the specific cell type and death pathway involved. Understanding these distinctions provides valuable insights into cellular fate and helps researchers and clinicians identify and interpret the significance of nuclear changes in various biological and pathological contexts.
As our understanding of cell death mechanisms continues to evolve, the study of these nuclear changes remains relevant for developing new therapeutic approaches and diagnostic techniques. Whether in basic research exploring fundamental cellular processes or in clinical pathology assessing disease states, the ability to recognize and interpret these nuclear alterations contributes significantly to our understanding of both normal physiology and pathological conditions.